10 mg/ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
NEFRISOL-P (Phenylephrine hydrochloride) is a vasoconstrictor and pressor drug chemically related to adrenaline and ephedrine. Chemically, Phenylephrine hydrochloride is designated as Benzenemethanol,3-hydroxy-α-[(methylamino)methyl]-,hydrochloride(R)-.(-)-m-Hydroxy-α-[(methylamino)methyl]benzyl alcohol hydrochloride. Its molecular formula is C9H13NO2·HCl and its molecular weight is 203.70.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
NEFRISOL-P is a clear, colourless sterile solution filled in 1 ml amber glass ampoule.
COMPOSITION :
Each ml contains :
Phenylephrine Hydrochloride I.P. 10 mg
Water for Injections I.P. q.s.
Contains no preservatives.
ACTIONS :
Phenylephrine hydrochloride produces vasoconstriction that lasts longer than that of adrenaline and ephedrine. Responses are more sustained than those to adrenaline, lasting 20 minutes after intravenous and as long as 50 minutes after subcutaneous injection. Its action on the heart contrasts sharply with that of adrenaline and ephedrine, in that it slows the heart rate and increases the stroke output producing no disturbance in the rhythm of the pulse. Phenylephrine hydrochloride is a powerful postsynaptic alpha-receptor stimulant with little effect on the beta-receptors of the heart. In therapeutic doses, it produces little if any stimulation of either the spinal cord or cerebrum. A singular advantage of this drug is the fact that repeated injections produce comparable effects.
The predominant actions of Phenylephrine hydrochloride are on the cardiovascular system. Parenteral administration causes a rise in systolic and diastolic pressures in man and other species. Accompanying the pressor response to Phenylephrine hydrochloride is a marked reflex bradycardia that can be blocked by atropine; after atropine, large doses of Phenylephrine hydrochloride increase the heart rate only slightly. In man, cardiac output is slightly decreased and peripheral resistance is considerably increased. Circulation time is slightly prolonged and venous pressure is slightly increased; venous constriction is not marked. Most vascular beds are constricted; renal splanchnic, cutaneous and limb blood flows are reduced but coronary blood flow is increased. Pulmonary vessels are constricted and pulmonary arterial pressure is raised. The drug is a powerful vasoconstrictor, with properties very similar to those of norepinephrine but almost completely lacking the chronotropic and inotropic actions on the heart. Cardiac irregularities are seen only very rarely even with large doses.
INDICATIONS :
NEFRISOL-P is intended for the maintenance of an adequate level of blood pressure during spinal and inhalation anaesthesia and for the treatment of vascular failure in shock, shock-like states and drug induced hypotension or hypersensitivity. It is also employed to overcome paroxysmal supraventricular tachycardia, to prolong spinal anaesthesia and as a vasoconstrictor in regional analgesia.
Administration :
NEFRISOL-P is generally injected subcutaneously, intramuscularly, slowly intravenously or in dilute solution as a continuous intravenous infusion. In patients with paroxysmal supraventricular tachycardia and if indicated, in case of emergency, NEFRISOL-P is administered directly intravenously.
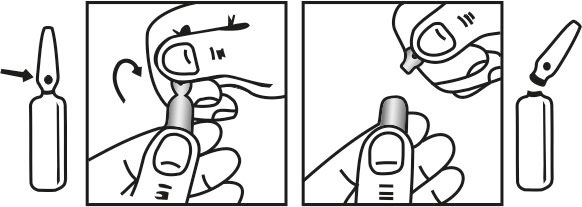
INSTRUCTIONS FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C. (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown.
Dosage :
The dose should be adjusted according to the pressor response.
Dosage calculations are shown in Table 1.
Table 1 – NEFRISOL- P Dosage and Administration
-Table-1.jpg)
-Table-2.jpg)
Use in Pregnancy :
Animal reproduction studies have not been conducted with Phenylephrine hydrochloride. It is also not known whether Phenylephrine hydrochloride can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Phenylephrine hydrochloride should be given to a pregnant woman only if clearly needed.
Use in Lactation :
It is not known whether this drug is excreted in human milk. Because many are excreted in human milk, caution should be exercised when Phenylephrine hydrochloride is administered to a nursing woman.
Use in Labour :
If vasopressor drugs are either used to correct hypotension or added to the local anaesthetic solution, the obstetrician should be cautioned that some oxytocic drugs may cause severe persistent hypertension and that even a rupture of a cerebral blood vessel may occur during the postpartum period.
Paediatric Use :
To combat hypotension during spinal anaesthesia in children, a dose of 0.5 mg to 1 mg per 25 pounds body weight, administered subcutaneously or intramuscularly, is recommended.
INTERACTIONS :
Vasopressors, particularly metaraminol, may cause serious cardiac arrhythmias during halothane anaesthesia and therefore should be used only with great caution or not at all.
MAO Inhibitors :
The pressor effect of sympathomimetic pressor amines is markedly potentiated in patients receiving monoamine oxidase inhibitors (MAOI). Therefore, when initiating pressor therapy in these patients, the initial dose should be small and used with due caution. The pressor response of adrenergic agents may also be potentiated by tricyclic antidepressants.
SIDE EFFECTS :
Headache, reflex bradycardia, excitability, restlessness and rarely arrhythmias.
OVERDOSAGE :
Overdosage may induce ventricular extrasystoles and short paroxysms of ventricular tachycardia, a sensation of fullness in the head and tingling of the extremities. The oral LD50 in the rat is 350 mg/kg, in the mouse 120 mg/kg.
TREATMENT OF OVERDOSAGE :
Should an excessive elevation of blood pressure occur, it may be immediately relieved by an a-adrenergic blocking agent, e.g. phentolamine.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C, protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
NEFRISOL-P is supplied as 10 mg Phenylephrine hydrochloride in 1 ml aqueous solution. Each 1 ampoule is packed in a carton with package insert. Such 10 cartons are packed in a outer carton.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.
 Cardiovascular
Cardiovascular